The genetic characteristics of a cancer vary from one patient to the next. So, even patients with the same type of cancer may respond differently to the same treatment, since their tumors have a unique compilation of genetic mutations that define their cancer.
Advances in molecular profiling technologies give physicians the ability to tailor treatment plans by analyzing DNA, RNA, and protein in a patient’s cancer cells to uncover information about the cancer that can inform treatment decisions. This approach helps in determining the right drug, for the right patient, at the right time.
DNA is the set of instructions used to make molecules called messenger RNAs which are translated into proteins. When DNA is mutated, it causes changes in proteins that can support tumor growth, cancer cell survival, or resistance to cancer treatment.
Understanding the mutations in a tumor’s DNA can provide valuable insight to help oncologists determine personalized treatments for each patient. By targeting the genetic changes driving tumor behavior, personalized treatment offers the opportunity to not only increase the efficacy of a therapy by targeting cancer cells but also decrease the possibility of administering treatments that may not be as beneficial to a specific patient.
What is next generation sequencing (NGS)?
Next-generation sequencing (NGS) is a broad term used to describe a number of modern genetic sequencing technologies. These advancing technologies allow researchers to analyze large amounts of DNA and RNA much more quickly and affordably than previous approaches, and have revolutionized the approach to personalized cancer care.
NGS provides a 'genetic survey' of a tumor tissue sample by identifying genetic alterations driving cancer. Researchers and clinicians now utilize NGS to identify specific changes in DNA by rapidly sequencing multiple genes within multiple samples to get a full understanding of the cancer.
There are two different types of molecular profiling tests – HotSpot Profiling and Comprehensive Profiling. Where HotSpot Profiling looks at specific locations within a gene known to be associated with cancer, Comprehensive Profiling looks at an entire gene to pinpoint all possible alterations that may be driving the cancer’s development and growth. Sarah Cannon has partnered with best-in-class commercial molecular profiling laboratories to give our physicians access to the best tests for our patients.
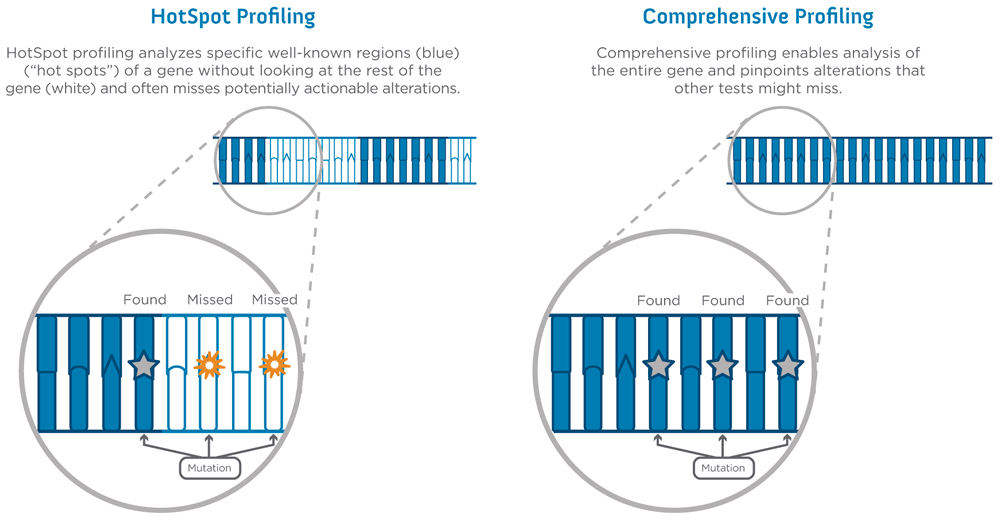
*HotSpot tests can be beneficial for identifying known alterations in specific genes that impact cancer growth and development. Comprehensive profiling expands on HotSpot testing to analyze both coding and non-coding regions of the DNA to give a more comprehensive view of the genes.
Types of Genetic Alterations

This type of mutation results from a change in one DNA base pair that leads to the substitution of one amino acid for another in the protein made by the gene. For example, the KRAS G12D mutation in colorectal cancer is a missense mutation caused by the substitution of DNA base pair G with A resulting in a change in the amino acid sequence of the protein from a glycine to an aspartate at codon 12 of KRAS.
Normal DNA: GCTGGT
Mutation: GCTGAT

An insertion occurs when genetic material is inserted into a region of DNA. For example, EGFR exon 20 insertions account for approximately 4–9.2% of all EGFR mutations in lung tumors.

A deletion occurs when genetic material is removed or deleted. Small deletions may remove one or a few base pairs, while larger deletions can be a complete or partial loss of a chromosome. For example, EGFR exon 19 deletions occur in approximately 48% of EGFR mutant lung tumors.

Chromosomal rearrangements are structural changes in chromosomes that result in genetic material being exchanged between chromosomes. Rearrangements may result in the production of a gene fusion that produces a malfunctioning proteins such as the Bcr/Abl fusion protein in chronic myeloid leukemia (CML). Chromosomal rearrangements can be balanced, with an even exchange of material with no genetic information gained or deleted between chromosomes, or unbalanced, where the exchange of chromosome material results in extra or missing genetic information.